ISO 10993-1 requires manufacturers to evaluate reasonably foreseeable misuse when determining biological safety requirements for a medical device. This concept recognizes that devices may not always be used exactly as intended, and biological risks must be evaluated for realistic use conditions that could occur in clinical practice.
In biocompatibility planning, this means considering whether variations in device use could change patient exposure, contact duration or affected tissues. These potential scenarios must be evaluated when defining the Biological Evaluation Plan (BEP) and selecting appropriate biological endpoints.
What Is Reasonably Foreseeable Misuse?
Reasonably foreseeable misuse refers to predictable patterns of device use that differ from the intended instructions but may still occur during real-world clinical use.
This concept is not meant to capture rare or purely hypothetical situations. Misuse scenarios must be supported by evidence, such as:
- Post-market surveillance (PMS) data
- Clinical literature
- User feedback and complaint data
- Risk management analysis under ISO 14971
The goal is to identify realistic use variations that could influence biological safety.
Examples:
Misuse scenarios may influence how a device interacts with the body and therefore may affect biocompatibility requirements.
- Extended use duration
A device intended for short contact may occasionally remain in place longer than planned. - Different tissue contact
A device designed for one tissue type may inadvertently contact another biological surface. - Use in different patient populations
Devices may be used in patients with different physiological characteristics or sensitivities. - Repeated or intermittent use
Intermittent use over long periods can result in a cumulative exposure duration that falls into a different regulatory contact category.
When such conditions are reasonably foreseeable, they should be incorporated into the biological evaluation strategy.
Impact on Biological Evaluation Planning
Considering foreseeable misuse can affect several aspects of biological evaluation, including:
- Device contact classification
- Contact duration category
- Selection of biological endpoints
- Exposure assumptions used in toxicological risk assessment
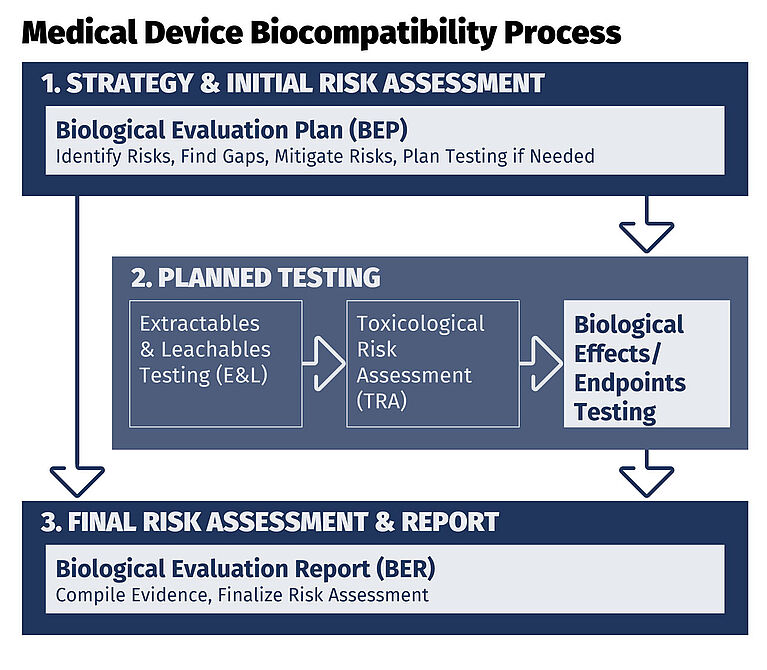
In practice, these scenarios may lead to additional justification, alternative testing strategies or expanded risk evaluation within the BEP and Biological Evaluation Report (BER).
Evidence-Based Risk Assessment
Regulators expect misuse scenarios to be systematically evaluated and documented. The analysis should demonstrate how the manufacturer determined whether a misuse scenario could affect biological safety.
When properly addressed, this process strengthens the scientific justification of the biological evaluation and supports transparent regulatory review.